Nonlocal Continuum Models for Oscillatory Media: Dynamics of Phase Equations
Recent experimental developments in biology and neuroscience allow researchers to record dynamical behavior from large spatial regions at high temporal resolution. Because this activity is often oscillatory, we can represent it with spatiotemporal phase maps that use phase equations to naturally model the dynamics. But despite a great deal of progress in the analysis of networks that comprise coupled phase oscillators (and recent generalizations such as higher-order phase models), the dynamics of spatially coupled phase models have seen limited results — particularly when the coupling is nonlocal.
Given this shortage, our group has been studying the dynamics of continuum systems of phase models in the form
\[\frac{\partial\theta(x,t)}{\partial t} = \omega(x) + \int_D W(|x-y|)H(\theta(y,t)-\theta(x,t))\enspace dy, \tag1\]
Here, \(x \in D\) and \(D\) is a one-dimensional (1D) or two-dimensional domain, \(W(|x|)\) determines the way in which interactions decay with distance, \(\omega(x)\) represents frequency heterogeneities, and \(H(\phi)\) is the phase interaction function (e.g., \(\sin(\phi)\) in the Kuramoto model). By varying the choice of domain shape and other parameters in \((1),\) we can explain a variety of spatiotemporal patterns in biological data while simultaneously developing new mathematical tools for their analysis.
![<strong>Figure 1.</strong> Examples of spatially organized oscillations in biological systems. <strong>1a.</strong> Local field potential recordings from a grid of implanted electrodes on a human cortex at two different epochs. <strong>1b.</strong> Cellular calcium activity across the proximal colon of a mouse indicates organized, small-amplitude ripple contractions. Figure 1a adapted from [1] and 1b courtesy of the author.](/media/apvfafnw/figure1.jpg)
We are especially curious about the existence of phase-locked solutions to \((1)\)—i.e., \(\theta(x,t) = \Omega t + \phi(x)\)—as well as their stability. We are also investigating the loss of phase locking as the frequency heterogeneities \(\omega(x)\) get too large, for example. Our interest in this class of models stems from our collaborations with several experimental groups that have successfully recorded large-scale oscillatory behavior (see Figure 1). Figure 1a illustrates the extracted phases from the local field potentials (LFPs) on a grid of implanted electrodes on a human cortex. First, the LFP is filtered, then a Hilbert transform is applied to extract the analytic phase. The top panel depicts a source, or bullseye, that propagates outward in a nearly radial wave; the bottom panel shows a counterclockwise rotating wave that is centered at the circled “core,” where the nearest neighbor phase differences are maximal. In Figure 1b, calcium activity from a piece of mouse colon shows that ripple contractions form traveling waves that move in the aboral to oral direction at regular intervals of about 10 cycles per minute. Patterns such as those in the top panel of Figure 1a and Figure 1b arise due to heterogeneities—i.e., in the local frequencies \(\omega(x)\)—while the rotating wave in the lower panel of Figure 1a results from a topological singularity.
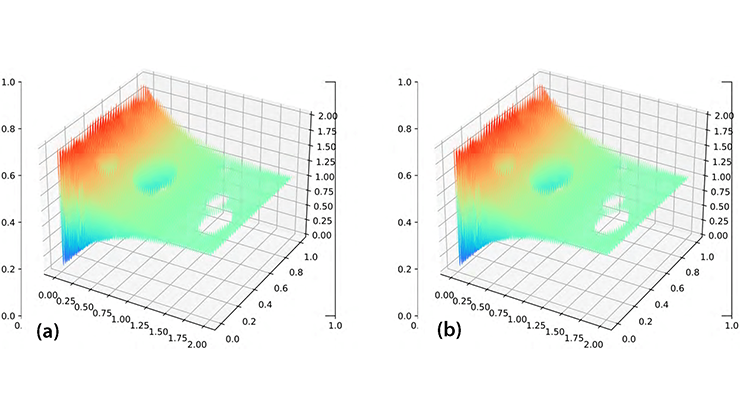
Modeling patterns like the ones in Figure 1a, where the apparent presence of a wave source leads to radially outward waves, is a relatively straightforward process. The top panel of Figure 2a depicts the steady state phase for \((1)\), where \(D\) is a square, \(W(x)\) is a Gaussian, \(H(\phi) = \sin(\phi+d)-\sin(d),\) and \(\omega(x)\) is a radially symmetric frequency gradient.
If we choose an annulus for domain \(D,\) we can then examine rotating waves of the form \(\theta(x,t) = \Omega t+\psi+f(r),\) where \(\psi\) is the polar angle and \(f(r)\) is an unknown function that satisfies a 1D integral equation [2]. The middle panel of Figure 2a is an example solution for the Gaussian \(W(x)\) and \(H(\theta) = \sin(\theta+d)-\sin(d)\) on an annulus with an outer diameter \(r=5\) and inner diameter \(r=1.\) When the hole shrinks, the solution fails to exist; in the absence of a hole, the “core” of the rotating wave produces asynchronous dynamics called a spiral chimera [4] (see the bottom panel of Figure 2a). As such, no true rotating waves appear in pure phase models without a central hole. Instead, the waves are broken up by disorganized activity at the core.
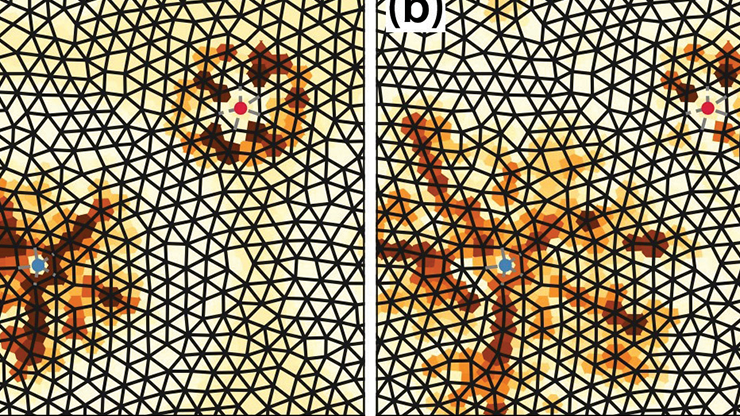
![<strong>Figure 2.</strong> Numerical simulations of \((1)\) in one-dimensional (1D) and two-dimensional domains. <strong>2a.</strong> The top panel depicts a square domain with a radially symmetric frequency gradient, where \(\omega(r)\) leads to outwardly propagating waves. The middle panel illustrates rotating waves in an annulus. Removal of the “hole” in the bottom panel triggers the emergence of a desynchronized core called a chimera. <strong>2b.</strong> The top panel portrays a 1D network with Gaussian connectivity and a linear frequency gradient. If the gradient is too large, as in the bottom panel, frequency plateaus arise. Top panel of Figure 2a and Figure 2b courtesy of Bard Ermentrout, middle and bottom panels of Figure 2a courtesy of [2].](/media/jo1d1if2/figure2.jpg)
A specialized class of spontaneously oscillating pacemaker cells called the interstitial cells of Cajal (ICCs) cause the oscillations in Figure 1b. The top panel of Figure 2b shows the results of a simulated 1D network of coupled phase oscillators when the coupling is Gaussian, a linear frequency gradient is present, and \(H(\phi)\) is taken from a mechanistic model for ICCs after the application of weakly coupled oscillator theory [3]. As expected, the resulting pattern consists of periodic waves that travel from \(x=1\) to \(x=0.\) In the late 1970s, John Neu explored a phase model approximation for a system of reaction-diffusion equations with weak diffusive coupling and found that the phase evolved as
\[\theta_t=\omega(x)+a{\theta^2_x}+b\theta_{xx}, \quad 0<x<1,\]
with appropriate boundary conditions [5]. As long as \(b>0,\) there will always be phase-locked solutions: \(\theta(x,t)=\Omega t+{\int^x_0}\phi(y)dy.\)
Our recent work considered phase-locked solutions to \((1),\) where \(D = [0,1]\) and \(W(x)\) is a symmetric Gaussian or exponential kernel with width \(\sigma\) [6]. By letting \(\sigma\) get small, we were able to explicitly establish phase-locked solutions away from the boundaries of the domain. In the reaction-diffusion case, locking always occurs regardless of the value of \(\omega(x);\) however, such an outcome is not true for \((1).\) The lower panel of Figure 2b utilizes the same model as the upper panel but increases the frequency gradient, which leads to a loss of locking and the emergence of so-called frequency plateaus: regions that are locked at the same frequency and separated by gaps. Three distinct plateaus are present in Figure 2b.
This brief article highlights multiple recent accounts of phase models that facilitate our understanding of the spatiotemporal organization of oscillatory media. Nonlocal coupling yields several new mathematical phenomena, such as spiral chimeras and frequency plateaus, that do not exist in continuum diffusion models. We look forward to continued advances in this area of study.
References
[1] Das, A., Zhang, J., Zabeh, E., Ermentrout, B., & Jacobs, J. (2025). Hidden spirals reveal neurocomputational mechanisms of traveling waves in human memory. Preprint, bioRxiv.
[2] Ding, Y., & Ermentrout, B. (2022). Rotating waves of nonlocally coupled oscillators on the annulus. SIAM J. Appl. Dyn. Syst., 21(3), 2047-2079.
[3] Ermentrout, G.B., & Kopell, N. (1984). Frequency plateaus in a chain of weakly coupled oscillators, I. SIAM J. Math. Anal., 15(2), 215-237.
[4] Martens, E.A., Laing, C.R., & Strogatz, S.H. (2010). Solvable model of spiral wave chimeras. Phys. Rev. Lett., 104(4), 044101.
[5] Neu, J.C. (1979). Chemical waves and the diffusive coupling of limit cycle oscillators. SIAM J. Appl. Math., 36(3), 509-515.
[6] Welsh, A.J., & Ermentrout, B. (2023). Mechanisms for producing oscillatory plane waves in discrete and continuum models. SIAM J. Appl. Math., 84(3), S151-S171.
About the Authors
Bard Ermentrout
Professor, University of Pittsburgh
Bard Ermentrout is a professor of mathematics at the University of Pittsburgh. He works in many areas of mathematical biology, with a focus on neuroscience.
Cameron Watt
Ph.D. student, University of Pittsburgh
Cameron Watt is a Ph.D. student in the Department of Mathematics at the University of Pittsburgh under the direction of Bard Ermentrout. His research focuses on dynamical systems.
Gavin Zhang
Ph.D. student, University of Pittsburgh
Gavin Zhang is a Ph.D. student in the Department of Mathematics at the University of Pittsburgh under the direction of Bard Ermentrout. His research focuses on dynamical systems.
Stay Up-to-Date with Email Alerts
Sign up for our monthly newsletter and emails about other topics of your choosing.