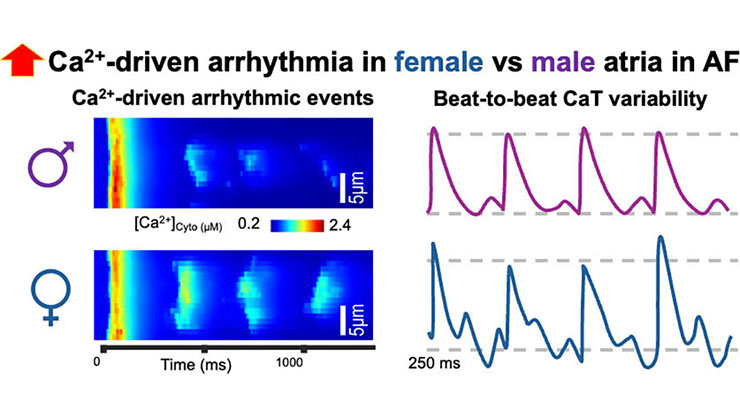
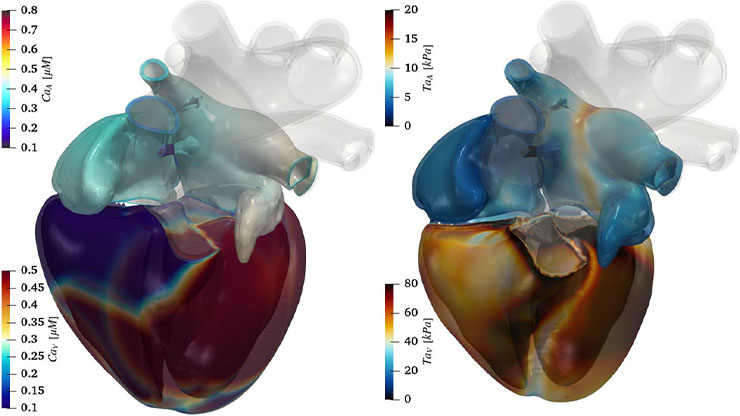
openCARP: Personalized Computational Model of the Heart Examines Cardiac Rhythm
What makes your heart beat? The answer may lie in advanced computational models that provide a deeper understanding of the fundamental mechanisms that drive the heartbeat or cause it to go awry — ultimately facilitating the development of personalized therapies to treat heart diseases. The openCARP software package, which uses cardiac bidomain equations to simulate the electrophysiology of the heart, marks a significant step forward in this important research area [6].
Atrial fibrillation is a heart rhythm disorder that originates in the atria (the heart’s upper chambers) and causes the heart to beat rapidly and irregularly. According to the Heart Rhythm Society, nearly 40 million people around the world experience this common condition, which is linked to poor sleep and daytime fatigue. Treatment is crucial to prevent stroke, heart failure, and other detrimental consequences, and tailored computer simulations can help identify optimal treatment pathways. For instance, individuals with atrial fibrillation can undergo a magnetic resonance imaging scan that enables the creation of a three-dimensional model of their heart. Specialists are then able to incorporate additional electrocardiogram (ECG) measures and adjust this computational model to reflect a patient’s unique heart function — eventually yielding a digital twin that allows cardiologists to identify precise targets for treatment procedures like ablation, which creates scar tissue in the heart that blocks the defective electrical signals that cause atrial fibrillation. Medical practitioners can also computationally simulate ablation to visualize a patient’s risk of developing new heart rhythm disorders [2]. If such risks are indeed detected, they adjust the ablation targets until an optimal therapy is reached. This innovative approach promises to enhance the precision and effectiveness of treatments for heart rhythm disorders, potentially improving outcomes and reducing risks for patients worldwide.
![<strong>Figure 1.</strong> Hierarchy of multiscale cardiac electrophysiology models, ranging from ion channels <strong>(1a)</strong> via integrated cell-level <strong>(1b)</strong> and tissue-level models <strong>(1c)</strong> to body surface and electrocardiogram measurements <strong>(1d)</strong>. The simulation system enables the investigation of “what-if” scenarios by changing the input parameters of the model (top row) and analyzing the subsequent effect on simulation outputs at numerous scales (bottom row) in comparison with experimental and clinical data. Figure courtesy of [4].](/media/kn5fe3cl/figure1.jpg)
The combination of numerical simulations and mathematical models of biophysical systems can improve our overall understanding of cardiac function. Because the human body is inherently a multiscale system, associated models must encompass spatial dimensions that range from nanometers to meters. The smallest components of many cardiac electrophysiology models are ion channels, which are only a few nanometers wide. These channels are integrated into cell models that are then combined into tissue models to study phenomena at the organ and body levels — including the optimization of atrial fibrillation therapy, assessment of the impact of specific gene mutations on the likelihood of heart rhythm disorders and their effects on ECG measurements, and optimal design of new pacemakers or cardiac catheters. This type of comprehensive numerical approach bridges the gap from the nanometer to meter scale (see Figure 1).
The openCARP software package [6] has emerged as a pivotal tool in the rapidly evolving field of cardiac electrophysiology simulations, setting new standards for the computational modeling and simulation of cardiac function. This open platform aims to facilitate cutting-edge research and enhance reproducibility and productivity in the scientific community. For added context, openCARP is a public-source cardiac electrophysiology simulation environment that enables in silico experiments and supports a wide range of users, including biomedical engineers, computational scientists, and specifically trained biologists and medical doctors. The software simulates cardiac electrophysiological processes at various scales, from the single-cell level to the entire heart. Its robust, flexible platform permits the creation and sharing of simulation pipelines, thereby automating in silico experiments and ultimately increasing reproducibility.
The package has already had a profound impact on scientific research, particularly in the realms of drug safety, diagnosis, risk stratification, therapy planning, and intraprocedural support. The software’s instrumental role in more than 150 peer-reviewed publications highlights its versatility and effectiveness in advancing our understanding of cardiac electrophysiology.
The following cardiac bidomain equations constitute the backbone of the openCARP simulator:
\[\begin{eqnarray}\nabla \cdot \sigma_i \nabla \phi_i &=& \beta I_m \\
\nabla \cdot \sigma_e \nabla \phi_e &=& {-}\beta I_m - I_e\\
I_m &=& C_m {{\partial V_m}\over {\partial t}} + I_{\textrm{ion}} - I_{\textrm{trans}}. \end{eqnarray}\]
These equations relate the intracellular electrical potential \(\phi_i\) to the extracellular electrical potential \(\phi_e\) through the transmembrane current density \(I_m\). Here, \(\sigma_i\) and \(\sigma_e\) are the intracellular and extracellular conductivity tensors, \(\beta\) is the ratio of cell membrane surface to tissue volume, \(C_m\) is the cell membrane capacitance per unit area, \(V_m\) is the transmembrane voltage \((\phi_i-\phi_e)\), and \(I_\textrm{ion}\) is the current density that flows through the ion channels in the cell membrane. Finally, \(I_\textrm{trans}\) is a transmembrane current density stimulus and \(I_e\) is an extracellular current density stimulus.
To solve these equations (or the simplified monodomain model), openCARP employs either PETSc (the Portable, Extensible Toolkit for Scientific Computation) or Ginkgo. Ginkgo offers high-performance linear algebra operations that are specifically optimized for graphics processing unit architectures and thus enhance computational efficiency [1]. Additionally, CVODE (part of Lawrence Livermore National Laboratory’s SUNDIALS suite) can expediate the solution of ordinary differential equations that arise in simulations of ionic currents and membrane voltages. All of these numerical backends collectively allow openCARP to perform detailed, accurate cardiac simulations that facilitate research and development in cardiac health. MICROCARD, a Centre of Excellence funded by EuroHPC, extends openCARP towards microstructural models with even higher resolution for deployment on large supercomputers.
Since its inaugural release in 2019, openCARP has undergone significant growth and development. The openCARP community has put together a comprehensive set of documentation and training materials, including 30 didactic hands-on examples and a suite of video tutorials. These resources are meant to help new users get started and empower experienced users to maximize the software’s potential. Regular user meetings, which take place both in person and online, foster community bonding and encourage knowledge sharing among users and developers. To date, the software’s codebase has seen over 5,000 commits from more than 30 contributors. Tailored infrastructure—including a searchable question-and-answer system, as well as tools and processes that ensure the long-term availability of citable software versions—promotes reproducibility and open science.
Sustainability is a central tenet of openCARP. The development team is committed to minimizing the required level of long-term maintenance and support through a comprehensive software management plan and extensive continuous integration, delivery, testing, and benchmarking pipelines. The openCARP package adheres to the FAIR Principles (findability, accessibility, interoperability, and reuse) for scientific data management and stewardship, which are essential for the promotion of open science. The software development team actively advocates for these principles and contributes to initiatives like the FAIR Principles for Research Software [3]. The integration of these ideas into the openCARP framework ensures the software’s continued value within the scientific community.
In recognition of its essential role in cardiac electrophysiology simulation, openCARP was honored with the Newcomer Prize of the first Helmholtz Incubator Software Award in 2024. Amir Jadidi, a clinical cardiologist at the Heart Center in Lucerne, Switzerland, is involved with several open-CARP-based research projects and praised its contributions to the broader cardiac community. “Computer models enhance our understanding of clinical observations and enable the transfer of anecdotal knowledge to other patients,” he said. “They are invaluable for systematically evaluating new algorithms that analyze clinical signals, as they can synthetically generate virtually any number of signals with known origins. This capability will become increasingly crucial as machine learning methods continue to proliferate.”
To conclude, we eagerly anticipate a future where simulations and prototyping accelerate the development of medical devices and personalized therapies. In silico methods provide quantitative, biophysical, and highly controlled approaches that have become integral to physiological and medical research. At the same time, computer simulations can complement biological experiments, animal testing, and clinical trials — thereby propelling the understanding of fundamental disease-causing processes and the creation of personalized therapies [5]. Mechanistic and data-driven modeling must synergistically work together to advance preventive, diagnostic, and therapeutic approaches. This integration of computer simulation and medical research promises to enhance the precision and effectiveness of new medical technologies, ultimately improving patient outcomes and furthering the field as a whole.
References
[1] Anzt, H., Cojean, T., Flegar, G., Göbel, F., Grützmacher, T., Nayak, P., … Quintana-Ortí, E.S. (2022). Ginkgo: A modern linear operator algebra framework for high performance computing. ACM Trans. Math. Softw., 48(1), 2.
[2] Azzolin, L., Eichenlaub, M., Nagel, C., Nairn, D., Sanchez, J., Unger, L., … Loewe, A. (2023). Personalized ablation vs. conventional ablation strategies to terminate atrial fibrillation and prevent recurrence. Europace, 25(1), 211-222.
[3] Chue Hong, N.P., Katz, D.S., Barker, M., Lamprecht, A.-L., Martinez, C., Psomopoulos, F.E., … RDA FAIR4RS WG. (2022). FAIR Principles for Research Software (FAIR4RS Principles) (Ver. 1.0). Zenodo. Retrieved from https://zenodo.org/records/6623556#.YqCJTJNBwlw.
[4] Loewe, A., Hunter, P., & Kohl, P. (2025). Computational modelling of biological systems now and then: Revisiting tools and visions from the beginning of the century. Philos. Trans. R. Soc. A. To be published.
[5] Loewe, A., Wülfers, E.M., & Seemann, G. (2018). Cardiac ischemia – insights from computational models. Herzschr. Elektrophys., 29(1), 48-56.
[6] Plank, G., Loewe, A., Neic, A., Augustin, C., Huang, Y.-L., Gsell, M.A.F., … Vigmond, E.J. (2021). The openCARP simulation environment for cardiac electrophysiology. Comput. Methods Programs Biomed., 208, 106223.
About the Authors
Axel Loewe
Group lead, Karlsruhe Institute of Technology
Axel Loewe leads the Computational Cardiac Modeling Group at Karlsruhe Institute of Technology in Germany. His research focuses on cardiac electrophysiology and biomechanics.

Aurel Neic
Chief executive officer and chief technology officer, NumeriCor GmbH
Aurel Neic is the chief executive officer and chief technology officer of NumeriCor GmbH in Austria. His research explores cardiac electrophysiology modeling and simulation, high-performance computing, and mesh generation.
Gunnar Seemann
Workshop trainer, soft skills coach, and team developer
Gunnar Seemann works as a self-employed workshop trainer, soft skills coach, and team developer for young academics. He previously served as a group leader for computational cardiology at Karlsruhe Institute of Technology and the University of Freiburg for 15 years.
Edward Vigmond
Department head, Liryc Heart Rhythm Disease Institute
Edward Vigmond is head of the modeling department at the Liryc Heart Rhythm Disease Institute in France. His research applies computer modeling to problems of cardiac electrophysiology and mechanics.
Gernot Plank
Professor, Medical University of Graz
Gernot Plank is a professor of computational cardiology at the Medical University of Graz in Austria. His research focuses on methods that investigate the multiscale-multiphysics mechanisms that underlie cardiac function in health and disease.

Related Reading
Stay Up-to-Date with Email Alerts
Sign up for our monthly newsletter and emails about other topics of your choosing.